Visual Inspection Defect Test Kits
Precision-manufactured particulate and cosmetic defect test kits for pharmaceutical visual inspection training, qualification, Knapp studies, and automated inspection system validation.

Our Capabilities
Comprehensive Visual Inspection Solutions
From custom defect test kits to surrogate formulations and full-service consulting, we support every stage of your visual inspection program.
Trusted Across the Industry
Serving Pharmaceutical & Biotech Organizations Nationwide
CDMOs
Contract Development & Manufacturing
Compounding Pharmacies
503A & 503B Facilities
Cell & Gene Therapy
Advanced Therapy Manufacturers
Biologics
Biopharmaceutical Companies
Large Pharma
Global Pharmaceutical Firms
Therapeutics
Injectable Drug Manufacturers
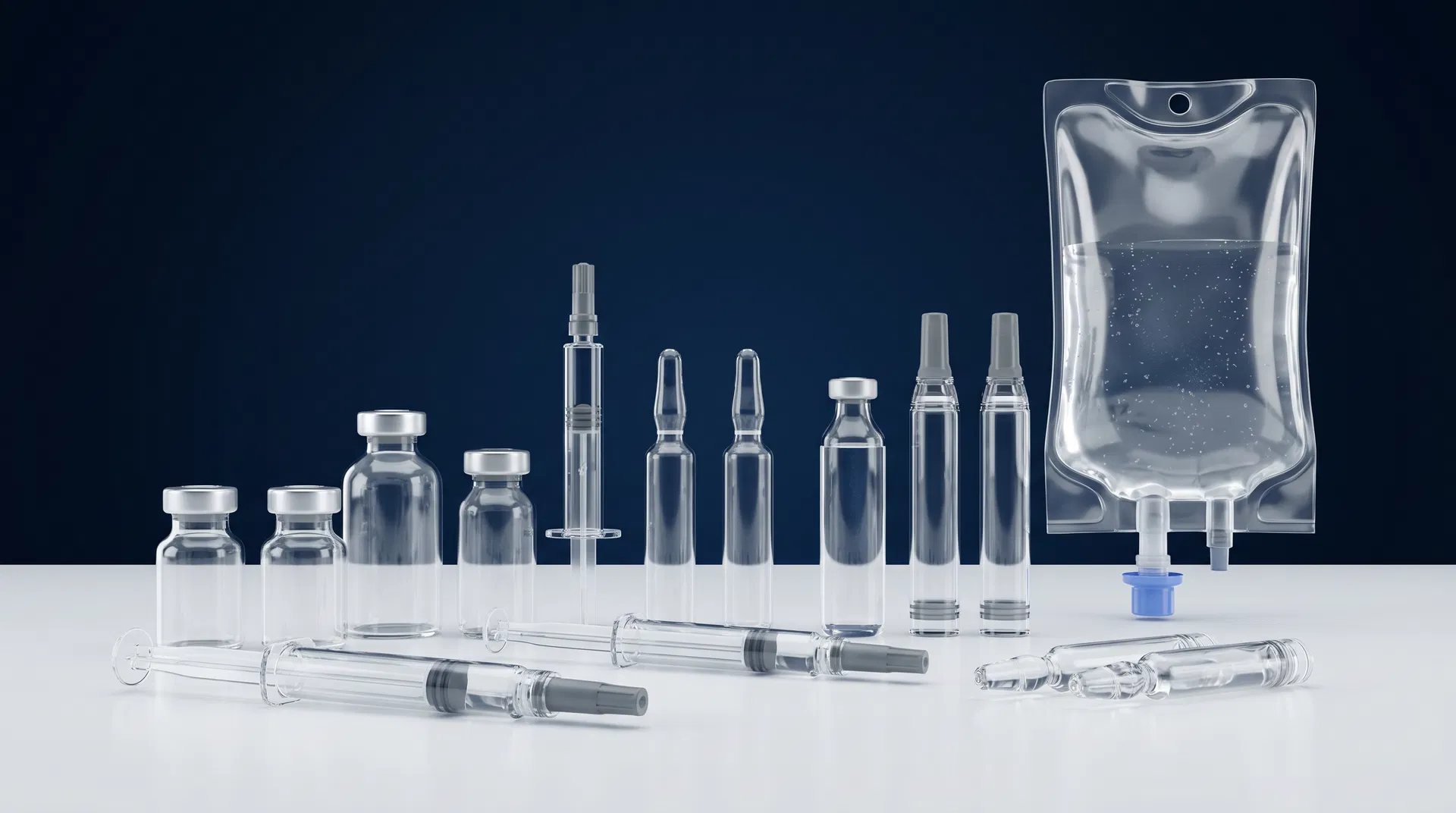
Any Container, Any Format
Defect Kits for Every Pharmaceutical Container Type
We manufacture particulate and cosmetic defect test kits for the full range of injectable containers. Our surrogate solutions can be customized to match your product's optical and physical properties — including index of refraction, density, and viscosity.

Why YB Inspection
Built for Regulatory Confidence
Every kit we produce is manufactured under a rigorous Quality Management System, fully traceable, and designed to meet the expectations of FDA, EU, and global regulatory bodies.
- 100% US-manufactured defect test kits
- Full QMS system — audit-ready at any time
- Custom surrogate solutions for any container type
- Compliant with FDA, USP <790>/<1790>, EU GMP Annex 1
- Support for vials, syringes, cartridges, bags, ampoules
- Automated filling solutions available
- Domestic shipping across the United States
- CGT, CDMOs, compounding pharmacies, biologics experience
Knowledge Hub
Technical Resources & Publications
We publish in-depth technical guides on visual inspection best practices, regulatory compliance, and equipment validation. Download our latest publications below.
Transitioning from Manual to Semi-Automated Visual Inspection (SAVI)
A comprehensive guide covering risk assessment, equipment qualification, operator training, and regulatory alignment for SAVI transitions per FDA and EU GMP requirements.
Establishing Compliant, Risk-Based Reject Limits for AVI Systems
A detailed framework for developing statistically sound, risk-based reject limits for automated visual inspection systems in compliance with FDA and ICH guidelines.
Ready to Elevate Your Visual Inspection Program?
Whether you need custom defect test kits, surrogate solutions, or expert consulting, we are here to support your quality objectives. Reach out today for a consultation.